
Chronic Obstructive Lung Disease:
Mr. Culver is 60 years old. He began smoking when he was 14. As a self-employed accountant, he has been able to smoke on the job his entire working life. He's estimated to have a 90-100 pack year history. Over the past week he has gotten increasingly short of breath and exercise intolerant and says maybe he's experienced some chills but he doesn't take his temperature so he doesn't know if he's been feverish. During the assessment interview Mr. Culver seems anxious and angry. He complains he can't breathe and he's "just ready for it to be over". He hasn't been able to sleep with is wife in 2 years because he sleeps upright in an easy chair and he suffers from sleep apnea. He and the wife used to enjoy meals out, but now food is a challenge. Sometimes it seems to take too much effort to chew and swallow. His food is always cold before he can get it down. On top of that his stomach is always in turmoil because he is often prescribed prednisone. He's on disability. His major accomplishments of the day are moving from the Barco-lounger to the bathroom and back and even that takes most of his energy. He doesn't know all the medications he's on because he doesn't care any longer. He also still smokes.
Mr. Culver is 60 years old. He began smoking when he was 14. As a self-employed accountant, he has been able to smoke on the job his entire working life. He's estimated to have a 90-100 pack year history. Over the past week he has gotten increasingly short of breath and exercise intolerant and says maybe he's experienced some chills but he doesn't take his temperature so he doesn't know if he's been feverish. During the assessment interview Mr. Culver seems anxious and angry. He complains he can't breathe and he's "just ready for it to be over". He hasn't been able to sleep with is wife in 2 years because he sleeps upright in an easy chair and he suffers from sleep apnea. He and the wife used to enjoy meals out, but now food is a challenge. Sometimes it seems to take too much effort to chew and swallow. His food is always cold before he can get it down. On top of that his stomach is always in turmoil because he is often prescribed prednisone. He's on disability. His major accomplishments of the day are moving from the Barco-lounger to the bathroom and back and even that takes most of his energy. He doesn't know all the medications he's on because he doesn't care any longer. He also still smokes.
COPD Pathophysiology
The lower airways of the lung are (in health) sterile. This is amazing when you think about how we constantly draw air into them that is full of toxins, particulate and pathogens. The pulmonary system can maintain this state of sterility due to numerous, overlapping redundant defense mechanisms. And smoking is hard on almost all of them.
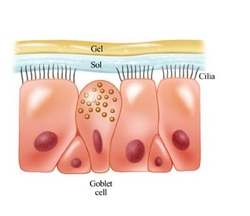
One of the principle defense mechanisms of the lung is the mucociliary escalator. From the trachea down to some of the smallest branches, airway epithelium is covered in specialized ciliated and mucous producing cells. Because of the physics of flow, the velocity of air drops off rapidly as it is inhaled into the numerous smaller branches. Any particles in the inhaled air decelerate and literally drop onto the sticky layer of mucous produced by goblet cells. Cilia beat rhythmically raising this mucous toward the mouth where it is swallowed as soon as it reaches the pharynx. So then this epithelium is both a physical barrier and a removal system for any harmful substance that might enter our lungs. The epithelium also produces bacteriocidal peptides ("defensins") in the event inhaled bacteria want to infect and prey on lung tissue. Deep in the lungs where there is no ciliated epithelium, the smallest airways and alveoli are populated with alveolar macrophages that phagocytize bacteria then mount inflammatory and immune responses to any infective organism that makes it down to the "business end" of the airways.
One of the principle defense mechanisms of the lung is the mucociliary escalator. From the trachea down to some of the smallest branches, airway epithelium is covered in specialized ciliated and mucous producing cells. Because of the physics of flow, the velocity of air drops off rapidly as it is inhaled into the numerous smaller branches. Any particles in the inhaled air decelerate and literally drop onto the sticky layer of mucous produced by goblet cells. Cilia beat rhythmically raising this mucous toward the mouth where it is swallowed as soon as it reaches the pharynx. So then this epithelium is both a physical barrier and a removal system for any harmful substance that might enter our lungs. The epithelium also produces bacteriocidal peptides ("defensins") in the event inhaled bacteria want to infect and prey on lung tissue. Deep in the lungs where there is no ciliated epithelium, the smallest airways and alveoli are populated with alveolar macrophages that phagocytize bacteria then mount inflammatory and immune responses to any infective organism that makes it down to the "business end" of the airways.

Tobacco smoke destroys much of the protective mechanisms that protect the lung. Acutely exposed to smoke, cilia on the epithelial layer are paralyzed allowing all the inhaled contaminants to sit on the lining of the airways. Chronic smoke exposure causes goblet cells to hypertrophy and produce abnormal, thick mucous in which cilia can't function and which is difficult to raise.
Tobacco smoke also subverts the cellular sentinels of the lung causing them to set up a slow, simmering, chronic inflammatory state. The alveolar macrophages that should be protecting the lung from infection begin producing proteinases that eat at and destroy the delicate architecture deep within the lung. During this inflammatory state, chemokines are elaborated that attract more and more inflammatory cells (principally neutrophils which bring more destructive enzymes and free radicals to destroy the tissues further.) Furthermore, the phagocytizing function of alveolar macrophages is destroyed by tobacco smoke making infection more likely if bacteria are inhaled deeply into the lung. All of these forces gradually destroy the extracellular matrix and the epithelial cells that should line the airways simply slough off. Abnormal "healing" processes initiated during this out-of-control inflammation only produce fibrosis and lung scarring.
Airway obstruction is also precipitated by tobacco smoke because it causes dilation of small blood vessels in the airway, makes them leaky and causes the inner layer to become edematous. Because of the perverse cell signalling set up by the toxins in tobacco smoke, the tissues of the airways undergo angiogenesis and engorgement which has profound mechanical effects in the smaller, deeper airways.
While destroying tissues deep in the lung, tobacco smoke also causes release of mediators that make airway smooth muscle contract. These smooth muscle cells eventually hypertrophy and irreversibly obstruct airflow in and (mostly) out of the lung. Tobacco smoke also causes the death of pneumocytes that produce surfactant predisposing the smallest airways to collapse further trapping air.
Tobacco smoke also subverts the cellular sentinels of the lung causing them to set up a slow, simmering, chronic inflammatory state. The alveolar macrophages that should be protecting the lung from infection begin producing proteinases that eat at and destroy the delicate architecture deep within the lung. During this inflammatory state, chemokines are elaborated that attract more and more inflammatory cells (principally neutrophils which bring more destructive enzymes and free radicals to destroy the tissues further.) Furthermore, the phagocytizing function of alveolar macrophages is destroyed by tobacco smoke making infection more likely if bacteria are inhaled deeply into the lung. All of these forces gradually destroy the extracellular matrix and the epithelial cells that should line the airways simply slough off. Abnormal "healing" processes initiated during this out-of-control inflammation only produce fibrosis and lung scarring.
Airway obstruction is also precipitated by tobacco smoke because it causes dilation of small blood vessels in the airway, makes them leaky and causes the inner layer to become edematous. Because of the perverse cell signalling set up by the toxins in tobacco smoke, the tissues of the airways undergo angiogenesis and engorgement which has profound mechanical effects in the smaller, deeper airways.
While destroying tissues deep in the lung, tobacco smoke also causes release of mediators that make airway smooth muscle contract. These smooth muscle cells eventually hypertrophy and irreversibly obstruct airflow in and (mostly) out of the lung. Tobacco smoke also causes the death of pneumocytes that produce surfactant predisposing the smallest airways to collapse further trapping air.
The Costs and Consequences of Chronic Tobacco Smoke Exposure
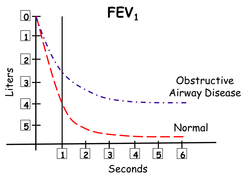
Once the architecture of the airways is destroyed and edema, thick mucous, and constricted smooth muscle characterizes all the airways, airflow is radically reduced, particularly during exhalation when intrathoracic pressure normally rises and presses on lung tissue. In addition, the amount of air that can be exhaled in a set period of time is reduced, leaving air behind (air trapping.) The lungs become chronically over-inflated.
This fact has profound effects upon on the work of breathing. An over-inflated lung-chest system is mechanically more difficult to expand further. Each inhalation comes at greater and greater cost energetically. Over-inflated lungs press down upon and flatten the diaphragm, (the principle muscle of respiration) causing muscle fibers to be shorter and less efficient. As a result, accessory muscles of respiration (SCM and trapezius) are employed to lift the chest wall out and up. In addition, the expiratory portion of the respiratory cycle should always be passive at rest. When patients have airway obstruction, the abdominal muscles are recruited to force air out of the lungs to make room for the next inhalation.
As a result of these anatomic and mechanical changes, air exchange in the alveoli is reduced. The sine qua non of alveolar hypoventilation is CO2 retention and chronic respiratory acidosis. This in turn results in compensatory hypercarbia (retention of HCO3- by the renal tubules). Hypoxia is also a natural result of the pathological changes in the lung. Oxygen diffuses 20 times more slowly than does CO2. It is not uncommon for patients with COPD to survive in the "50/50 Range". (PaO2 50 mmHg or less and PaCO2 50 mmHg or more.) In addition to the physics of oxygen diffusion coefficients, hypoxemia can be made worse by imbalances in ventilation and perfusion (V/Q mismatch). Under these conditions, hypoxemia is refractory.
This fact has profound effects upon on the work of breathing. An over-inflated lung-chest system is mechanically more difficult to expand further. Each inhalation comes at greater and greater cost energetically. Over-inflated lungs press down upon and flatten the diaphragm, (the principle muscle of respiration) causing muscle fibers to be shorter and less efficient. As a result, accessory muscles of respiration (SCM and trapezius) are employed to lift the chest wall out and up. In addition, the expiratory portion of the respiratory cycle should always be passive at rest. When patients have airway obstruction, the abdominal muscles are recruited to force air out of the lungs to make room for the next inhalation.
As a result of these anatomic and mechanical changes, air exchange in the alveoli is reduced. The sine qua non of alveolar hypoventilation is CO2 retention and chronic respiratory acidosis. This in turn results in compensatory hypercarbia (retention of HCO3- by the renal tubules). Hypoxia is also a natural result of the pathological changes in the lung. Oxygen diffuses 20 times more slowly than does CO2. It is not uncommon for patients with COPD to survive in the "50/50 Range". (PaO2 50 mmHg or less and PaCO2 50 mmHg or more.) In addition to the physics of oxygen diffusion coefficients, hypoxemia can be made worse by imbalances in ventilation and perfusion (V/Q mismatch). Under these conditions, hypoxemia is refractory.
Cardiovascular Consequences
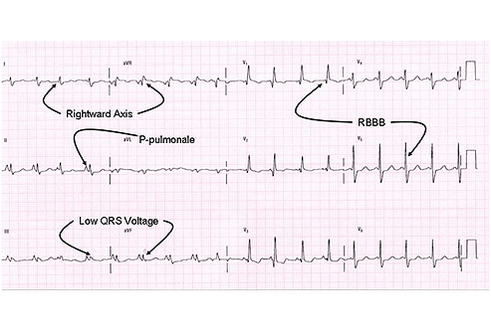
Tissue changes in the lung have profound consequences for the heart. Lung blood flow is normally shunted away from poorly oxygenated alveoli maintaining roughly balanced ventilation and perfusion. When there is organ-wide vasoconstriction (secondary to large areas of poor ventilation, the lung is no longer a high-volume low pressure circulatory system. The right ventricle must work harder to move blood into the lungs. It eventually hypertrophes. (Since the right atrium is the seat of the S-A node (natural pacemaker), it is not uncommon for patients with advanced COPD to have PAC's in addition to the changes in conduction characteristic of cor pulmonale.)
And One More Thing
COPD associated chronic hypoxemia causes the kidneys to produce extra amounts of erythropoitin. This, in turn, stimulates erythrocyte production in the bone marrow. Such secondary polycythemia is typical in patients with chronic bronchitis and is responsible for what is sometimes called a "plethoric and ruddy" appearance.
